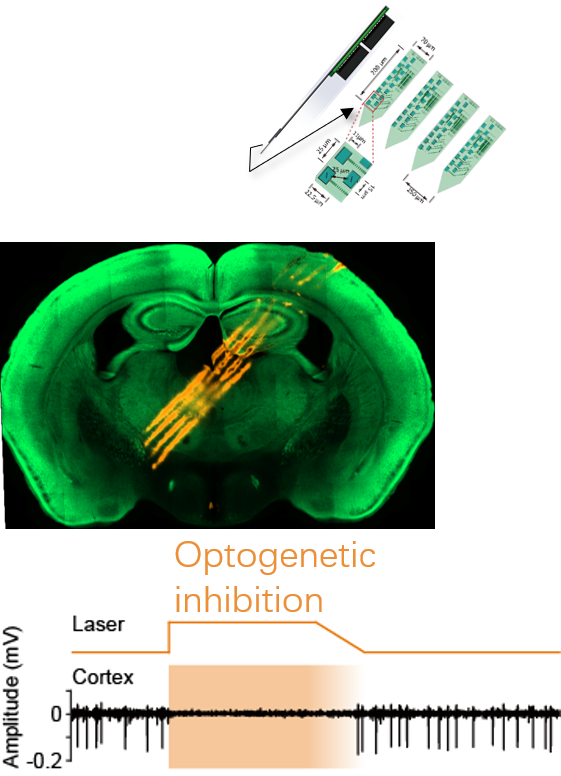
multichannel recording using silicon probes 
two photon calcium imaging(Ai93D x Emx1-cre; M2 area;)
The mammalian brains have millions to billions of neurons with trillions of connections. This complicated network supports numerous behaviors including high level cognitive functions such as working memory, decision-making.
Understanding how the neural circuits generate behaviors is a fundamental challenge in modern neuroscience. To understand how working memory is generated, maintained and discharged in the mammalian brain, we integrate optogenetics, multi-channel recording, 2-photon imaging and quantitative behavior to probe neural dynamics underlying working memory.